Atopic dermatitis (AD) is a chronic inflammatory disease defined by inherited or acquired defects in the skin barrier, which allow for the penetration of allergens and microbes. This dysfunction triggers a type 2-skewed immune response, creating a self-sustaining pathologic cycle where inflammation further degrades the stratum corneum. Crucially, these barrier defects and sub-clinical inflammation are present even in nonlesional skin, contributing to the “atopic march”—the sequential development of other allergic comorbidities like asthma and food allergies. Dupilumab, a monoclonal antibody that blocks the shared receptor for IL-4 and IL-13, was considered a potential solution because these cytokines are central drivers of type 2 inflammation. By blocking this pathway, researchers hypothesized that dupilumab could break the inflammatory cycle, repair the skin barrier, and potentially prevent the systemic “spill-over” of cytokines that leads to further allergic diseases.
Methods
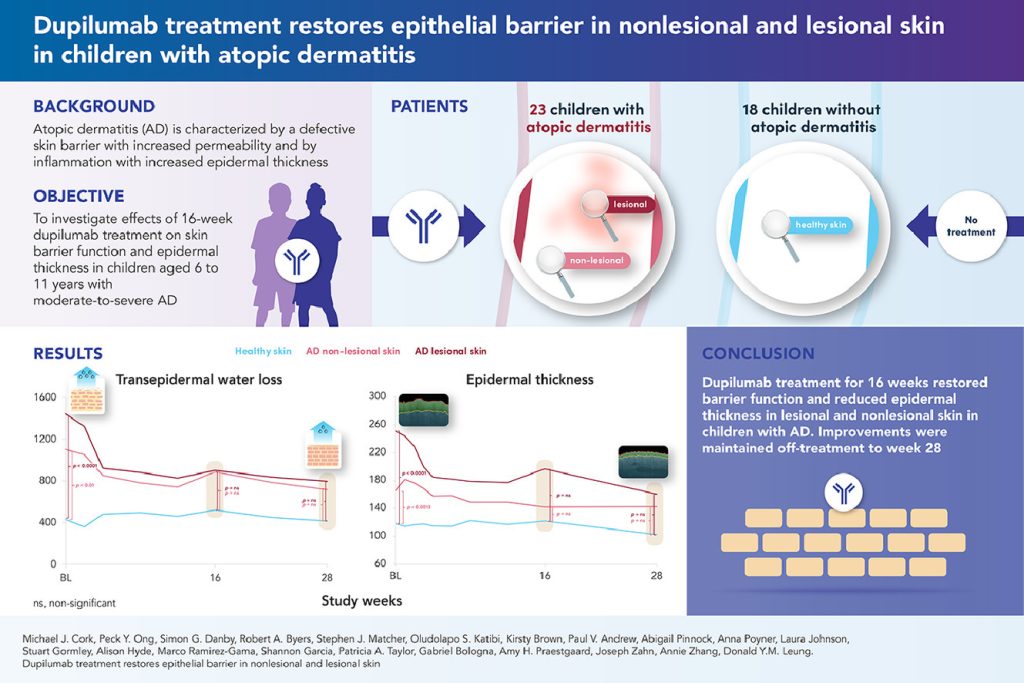
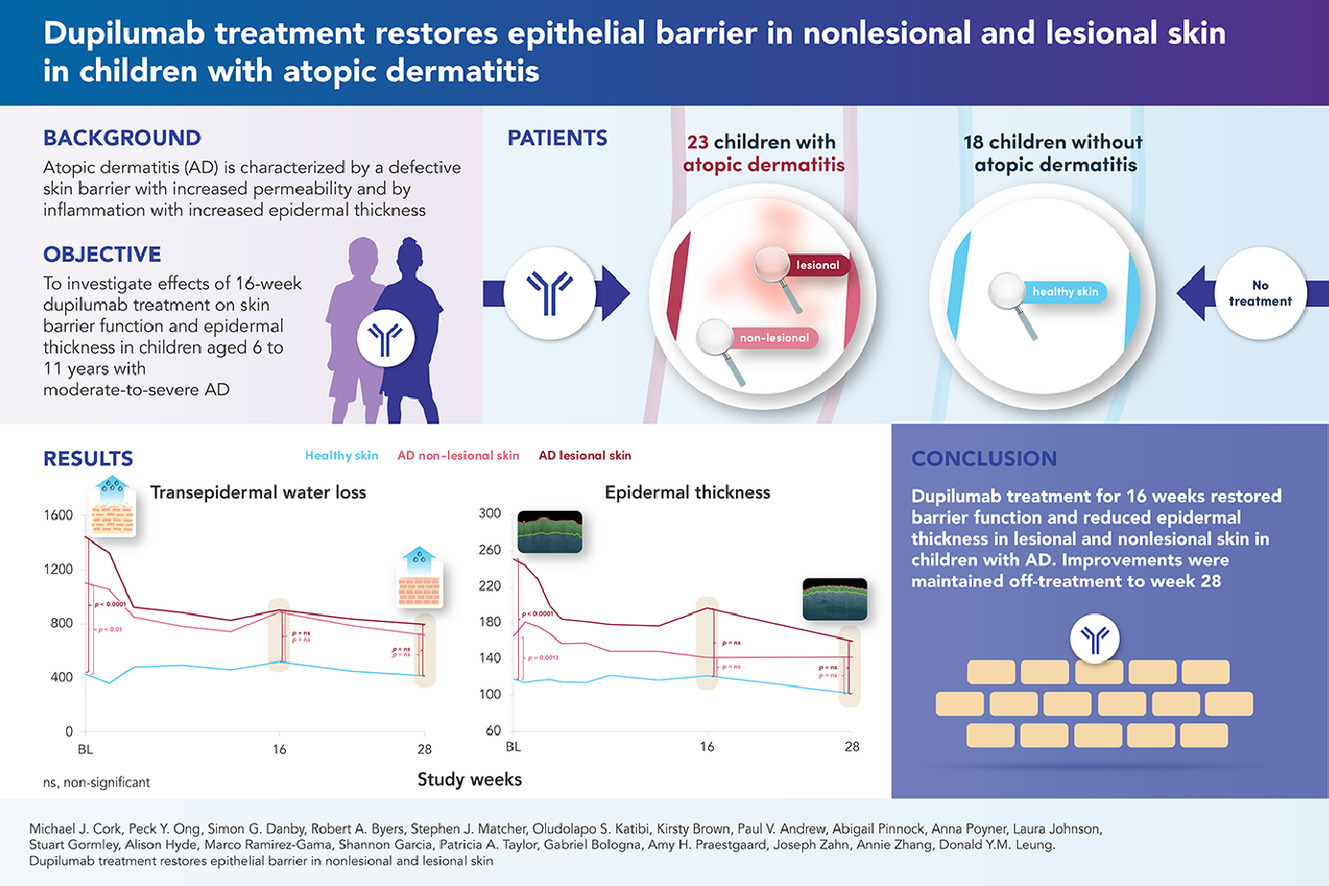
The PELISTAD study was an open-label, phase 4 trial involving 23 children (aged 6–11) with moderate-to-severe AD and 18 matched healthy volunteers. Participants received subcutaneous dupilumab for 16 weeks, followed by a 12-week observational period to assess sustained effects. Researchers utilized non-invasive techniques, including transepidermal water loss (TEWL) to measure barrier function and optical coherence tomography (OCT) to monitor epidermal thickness and inflammation in situ.
Key Findings
• Rapid Barrier Restoration: Treatment with dupilumab led to a significant reduction in basal TEWL in both lesional and nonlesional skin, with values becoming no longer significantly different from healthy skin by week 4.
• Improved Structural Integrity: Normalization of TEWL after skin tape-stripping (STS) suggests that dupilumab enables the skin to better withstand physical disruption.
• Reduction in Epidermal Hyperplasia: OCT imaging revealed that dupilumab significantly reduced epidermal thickness in both lesional and nonlesional areas, addressing sub-clinical inflammation that is often invisible to the naked eye.
• Clinical and Quality of Life Improvements: Significant decreases were observed in clinician-rated severity (EASI) and patient-reported outcomes, including itch and skin pain, by week 16.
• Sustained Efficacy: The improvements in skin barrier function and epidermal thickness achieved during the treatment period were maintained through the week 28 follow-up.
The novelty of this research lies in its demonstration that dupilumab restores the epithelial barrier and reduces epidermal thickness not only in visible lesions but also in clinically unaffected nonlesional skin in children. This provides objective, non-invasive evidence that systemic treatment can address sub-clinical skin inflammation. The future implications are profound: because childhood is a “window of opportunity” marked by high immune plasticity, early intervention with dupilumab may offer disease-modifying effects. By repairing the skin barrier early, it may be possible to halt the atopic march and prevent the development of lifelong allergic comorbidities.
Link to the study: https://www.sciencedirect.com/science/article/pii/S1081120626000281