The skin acts as a vital biological armor, protecting the body against environmental pollutants and pathogens that trigger inflammation. While inflammation is a necessary response to injury, chronic or excessive inflammation leads to the release of pro-inflammatory cytokines like IL−1α and TNF–α, which upregulate matrix metalloproteinases (MMPs) that degrade essential structural proteins such as collagen and elastin. This degradation impairs wound healing and compromises the epidermal barrier by disrupting tight junctions and desmosomal proteins. While glycerin is a staple cosmetic ingredient for hydration, it lacks significant regenerative or anti-inflammatory properties on its own. Ozonized glycerin (OG) was considered a potential solution because it stabilizes ozone—a powerful therapeutic agent with antibacterial and anti-inflammatory effects—into a safe, stable, and controlled-release format, effectively overcoming the toxicity and instability issues of gaseous ozone.
Methods
Researchers utilized 3D reconstructed human epidermis to evaluate wound closure over 13 days and ex vivo human skin biopsies to study inflammatory responses over a 4-day period. Tissues were pretreated with either 1% glycerin or 1% ozonized glycerin, with some explants challenged with lipopolysaccharides (LPS) to simulate an inflammatory environment. The study employed immunofluorescence staining to quantify structural proteins like collagen, elastin, and claudin-1, while ELISA assays measured the release of cytokines and growth factors.
Key Findings
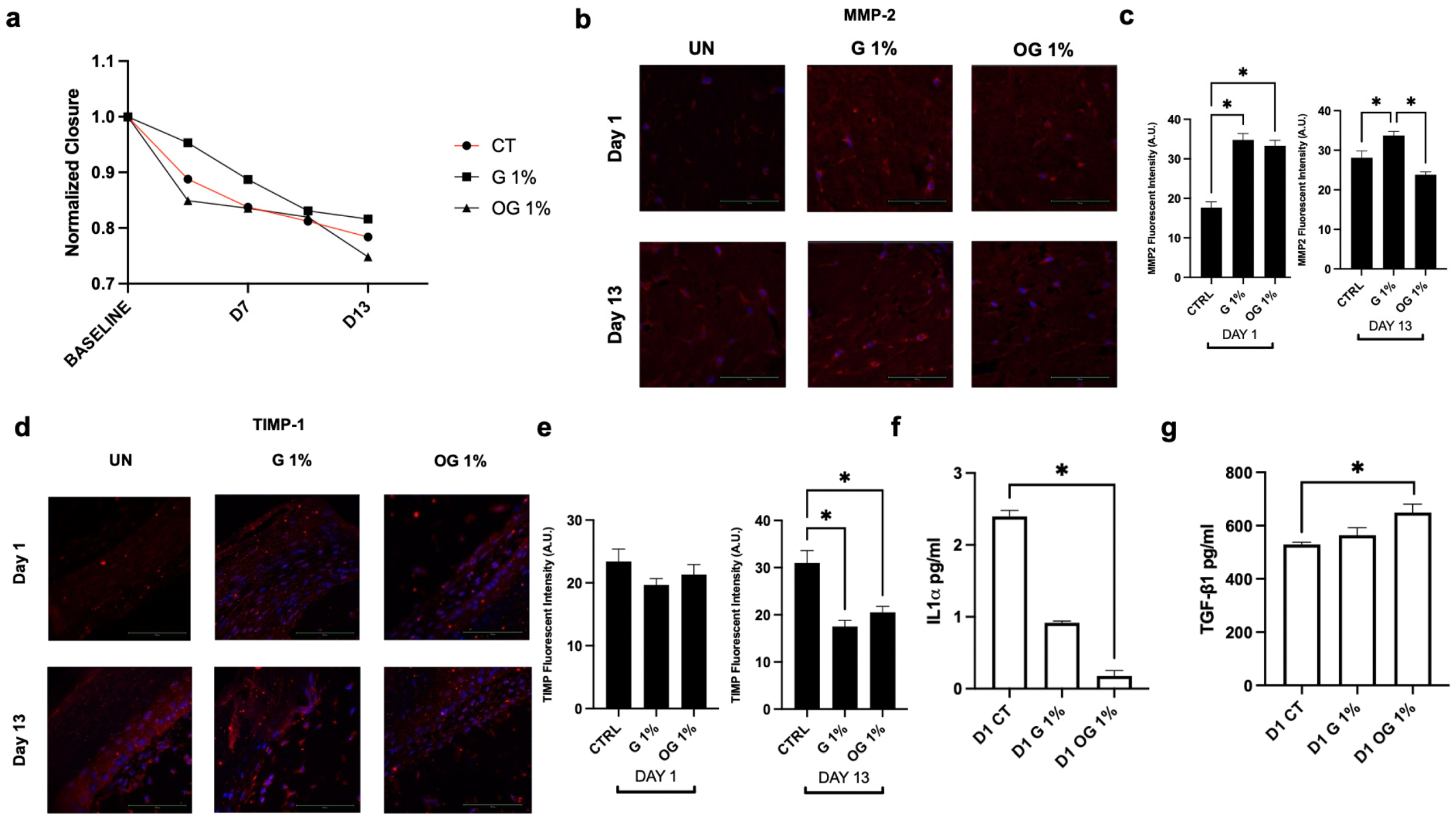
• Accelerated Wound Healing: OG treatment significantly enhanced wound closure by 6.8% compared to standard glycerin in the 3D epidermal model.
• Cytokine Modulation: Application of OG led to a significant reduction in pro-inflammatory IL−1α release while simultaneously promoting the secretion of the regenerative growth factor TGF-1β.
• Reinforced Skin Barrier: OG uniquely upregulated critical barrier proteins, including claudin-1 and desmocollin-1, which are essential for maintaining epidermal cohesion and integrity.
• Extracellular Matrix (ECM) Protection: In inflammatory models, OG protected against LPS-induced elastin degradation (showing a 67.7% difference from LPS-only groups) and reduced the expression of the degradative enzyme MMP-9.
• Enhanced Matrix Deposition: OG stimulated an environment favorable for ECM reinforcement by increasing collagen type III and TIMP-1 expression under basal conditions.
The novelty of this research lies in its first-time demonstration of ozonized glycerin’s ability to modulate tight junctions and the extracellular matrix within physiologically relevant 3D and ex vivo human skin models, specifically identifying the glycerin-derived peroxide 1,2,4-trioxepane as a key active species. These findings suggest that OG is a multifunctional cosmetic ingredient capable of enhancing skin regeneration and resilience beyond simple moisturization. Future implications include the development of advanced topical therapies for clinical dermatology; however, future studies should focus on in vivo models to capture the full complexity of healing and better understand the skin’s absorption and release of ozone-derived compounds.
Link to the study: https://www.mdpi.com/2079-9284/13/1/42
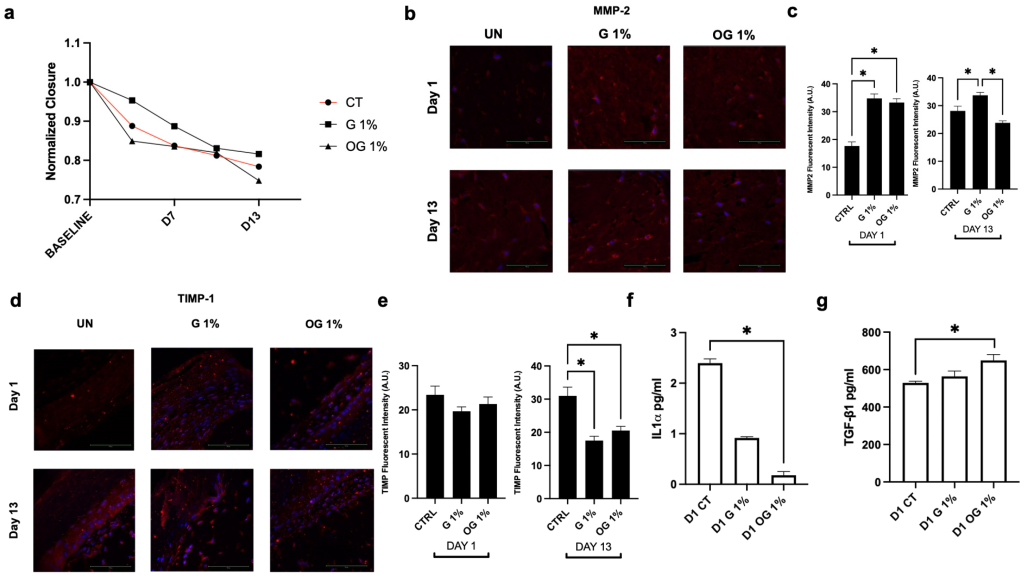
OG promotes wound closure and modulates remodeling and inflammatory markers in 3D skin tissues. (a) Wound closure quantified over a 13-day period. Representative immunofluorescent images of (b) MMP-2 and (c) quantification of protein expression levels at Day 1 and Day 13. Representative immunofluorescent images of (d) TIMP-1 and (e) quantification of protein expression levels at Day 1 and Day 13. (f) IL-1α and (g) TGF-1β release in culture medium after pretreatment with OG and G in the wound closure model at Day 1. Nuclei are counterstained with DAPI (blue). Data are presented as mean ± standard error of the mean. Statistical significance was determined using one-way analysis of variance with Tukey’s post hoc test for multiple comparisons (n = 3). A p-value < 0.05 was considered statistically significant and denoted by an asterisk (*).