Chronic and acute skin injuries, including burns and ulcers, represent a major global clinical challenge that places a significant socioeconomic burden on healthcare systems. While traditional cell-based therapies like stem cell grafting offer some relief, they are often hindered by limitations such as immune rejection, potential tumorigenicity, and difficulties in scaling production. Consequently, researchers have turned to cell-free regenerative approaches, specifically extracellular vesicles (EVs), which act as nanoscale messengers carrying bioactive cargo to facilitate intercellular communication and tissue repair. This study explores a novel source for these vesicles: Scandinavian planarian flatworms, known as “super-regenerators” for their ability to rebuild their entire bodies from small fragments. Because planarians utilize EV-mediated signaling to drive their own extraordinary regeneration, they were considered a potent potential source for cross-species (xenogeneic) therapeutic factors that might “re-engage” dormant repair programs in human tissue.
Methods
Researchers isolated EVs from wild-type Swedish planarians using collagenase-based tissue dissociation followed by sequential centrifugation and polyethylene glycol (PEG) aggregation. The resulting vesicles were characterized for size and morphology through dynamic light scattering and cryo-transmission electron microscopy. Therapeutic efficacy was subsequently evaluated using a chicken chorioallantoic membrane (CAM) burn model and a human 3D skin equivalent for mechanical puncture wounds. Finally, the immunogenicity of the EVs was assessed by exposing them to human primary peripheral blood mononuclear cells.
Key Findings
- Accelerated Wound Closure: Treatment with planarian EVs significantly speedup tissue regeneration and wound closure in both the avian burn model and human 3D skin puncture model compared to controls.
- Improved Skin Integrity: The EVs enhanced re-epithelialization and barrier integrity, with topically applied vesicles proving particularly effective in promoting the outgrowth of the epidermis into wound regions.
- Fibroblast Proliferation: Adding EVs to the culture medium resulted in a significant increase in the thickness of the fibroblast-rich bottom layer of human skin models, suggesting a potential application for treating age-induced skin thinning.
- Vesicle Stability and Morphology: Characterization confirmed the presence of stable, lipid bilayer-enclosed vesicles with a mean diameter of 90 nm, which remained functional even after 16 days of storage at 4 °C.
- Low Immunogenicity: Preliminary tests on human primary immune cells showed no significant adverse effects on cell count or activity, indicating the potential for safe cross-species application.
This research is highly novel as it represents the first demonstration of an EV-based intervention derived from a highly regenerative invertebrate applied to mammalian wound models. By proving that planarian-derived factors can successfully orchestrate repair across species, the study bridges a significant knowledge gap in regenerative medicine. The future implications of this work are vast, positioning these vesicles as a novel, cell-free therapeutic strategy for treating difficult clinical cases such as chronic burns and non-healing ulcers, while also offering a potential pathway for cosmetic skin rejuvenation.
Link to the study: https://pubs.acs.org/doi/full/10.1021/acsomega.5c11592
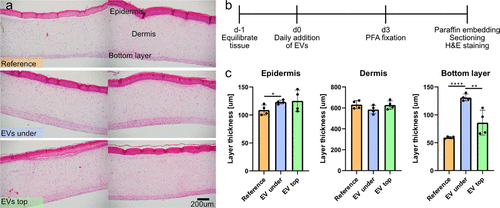
In the image: Pristine human artificial skin samples. (a) Optical micrographs showing H&E stained sections with the different layers indicated. The skin samples were cultured as recommended (“reference”), with EVs added to the culture medium (“under”) or with EVs on top of the epidermis (“top”). (b) Timeline depicting the outline of the experiment. (c) Thickness of the different layers. Two biological and two technical replicates per condition. Stars denote statistical significance: * p = 0.026, **** p < 0.0001, ** p = 0.0081.
