Atopic dermatitis (AD) is a chronic inflammatory skin condition characterized by eczematous lesions and intense itching, which imposes a significant physical, psychological, and economic burden on patients and their caregivers. While mild cases are typically managed with topical treatments and emollients, moderate-to-severe cases often require systemic immunomodulatory agents. Dupilumab was considered a potential solution because it is a monoclonal antibody that targets the IL-4Rα subunit, effectively blocking the IL-4 and IL-13 signaling pathways that drive type 2 inflammation. This targeted mechanism offers rapid clinical benefits and a superior safety profile compared to traditional systemic therapies.
Methods
This retrospective longitudinal study followed 61 patients with severe atopic dermatitis (EASI ≥ 24) treated with dupilumab over 12 months. Clinical outcomes were evaluated at baseline, 16 weeks, and 52 weeks using physician-reported Eczema Area and Severity Index (EASI) scores. Additionally, patients provided input through various Patient-Reported Outcome Measures (PROMs), including the Atopic Dermatitis Control Tool (ADCT), Dermatology Life Quality Index (DLQI), and Numerical Rating Scales (NRS) for itch and sleep.
Key Findings
- Significant Initial Improvement: At 4 months, 59% of patients reached the physician-targeted EASI score of ≤ 7, and 50.8% achieved adequate disease control via ADCT.
- Long-term Efficacy: By 12 months, the proportion of patients “in control” rose to 85.2% for EASI scores and 83.6% for ADCT scores.
- Enhanced Quality of Life: At the one-year mark, 86.9% of patients reached a DLQI score of ≤ 5, and 91.8% achieved a sleep-NRS score of ≤ 4.
- Meaningful Clinical Change: Every patient (100%) achieved a minimally clinically important difference in their ADCT score by the end of the 12-month study period.
- Safety Profile: Side effects were limited to mild conjunctivitis in 21.3% of the cohort, none of which necessitated the discontinuation of therapy.
The novelty of this research lies in its use of both physician-assessed data and a comprehensive suite of patient-centered tools—most notably the Atopic Dermatitis Control Tool (ADCT)—to evaluate long-term disease control in a real-world setting. The study demonstrates that while many patients see rapid benefits, a full 12 months of therapy is often required to meet “treat-to-target” goals. The future implication of these findings is that the ADCT could serve as a versatile, primary tool for clinicians to monitor treatment outcomes, potentially replacing the need for multiple, separate patient-reported questionnaires in routine practice.
Link to the study: https://journals.lww.com/ijd/fulltext/9900/evaluation_of_outcomes_of_dupilumab_therapy_in.284.aspx
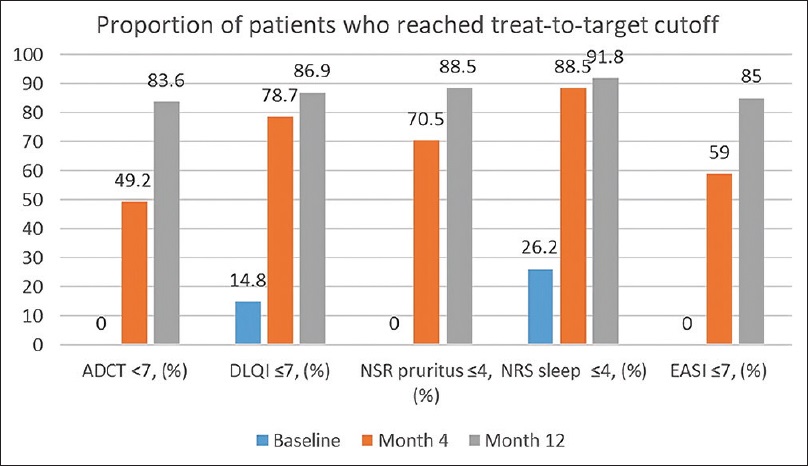
In the figure: Disease control according to different treat-to-target cutoffs at baseline, 4 and 12 months follow-up
