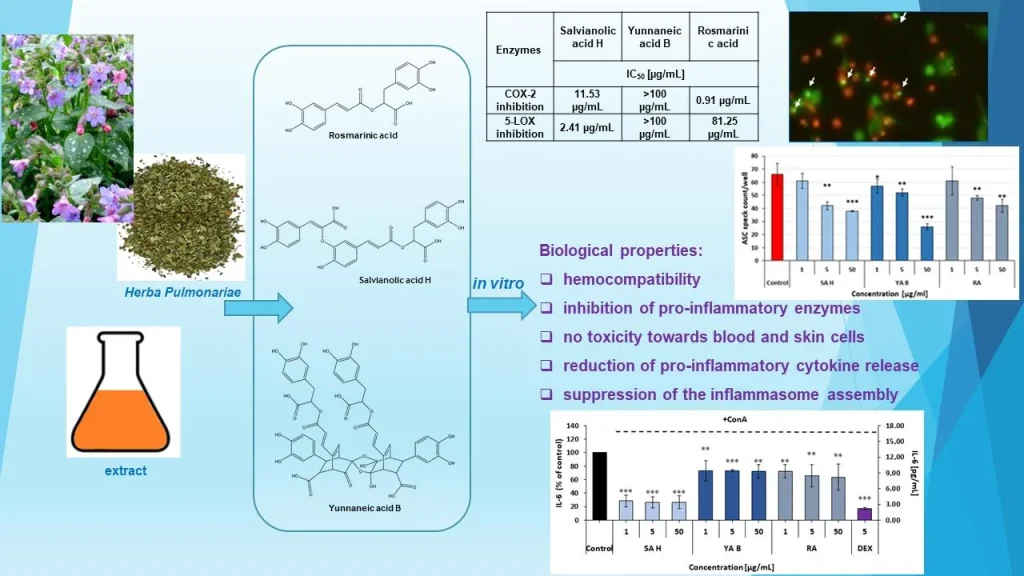
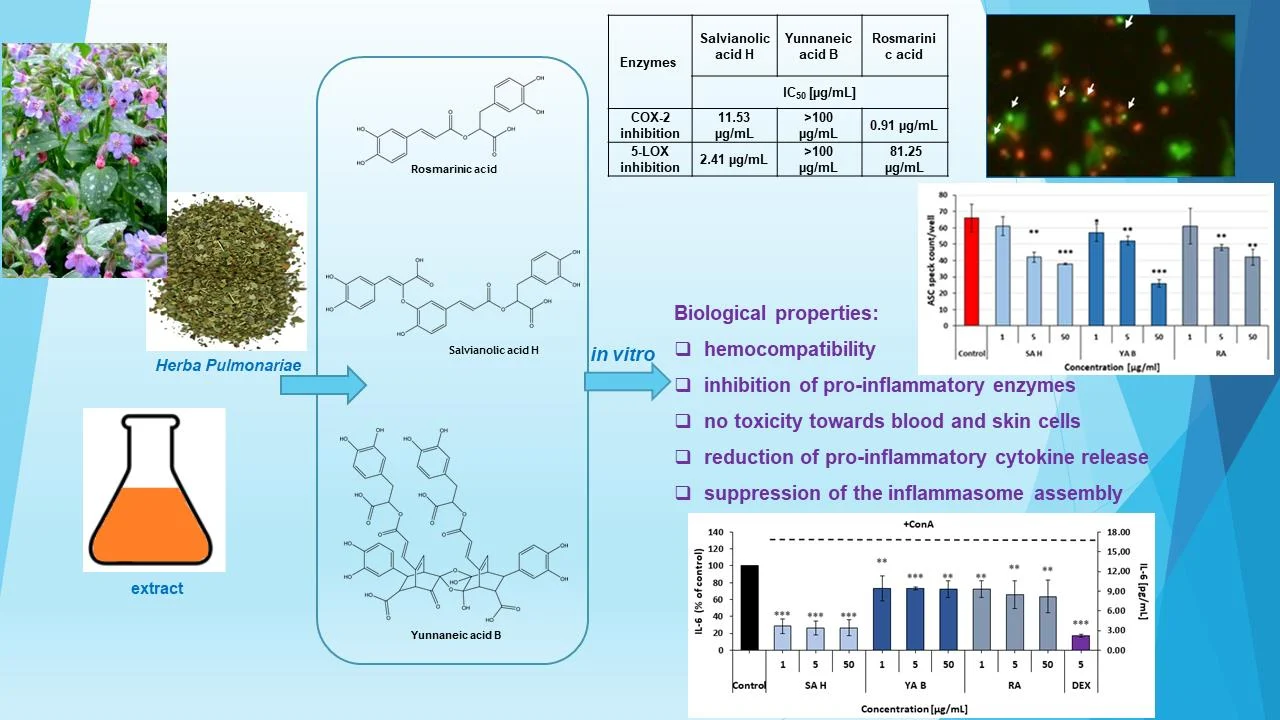
The physiological process of wound healing is a complex progression that involves three main phases: initiation, controlled development, and tissue recovery. While inflammation is an integral part of this process, excessive or prolonged (chronic) inflammation can lead to tissue damage, increased scarring, and a disruption in the healing process. Traditional solutions often involve phenolic acids, such as rosmarinic acid (RA), which are well-recognized for their antioxidant and anti-inflammatory properties in pharmaceuticals and cosmetics. However, the therapeutic potential of its more complex oligomeric derivatives, such as salvianolic acid H (SA H) and yunnaneic acid B (YA B), remained largely uncharacterized despite evidence that their larger molecular mass and distinct structures might offer enhanced biological effects or unique interactions with cell membranes. This study considered these derivatives as potential answers to the need for more effective topical agents that can suppress uncontrolled inflammatory responses and improve wound-healing outcomes.
Methods
This study comparatively evaluated the anti-inflammatory potential and hemocompatibility of SA H and YA B against their parent compound, RA, using human skin cell lines (HaCaT and NHDF), monocytes, and PBMCs. Researchers utilized ELISA to measure cytokine release, live-cell imaging to monitor NLRP3 inflammasome formation, and specialized kinetic assays to assess blood plasma coagulation and fibrinolysis. Enzyme inhibition was screened for COX-2 and 5-LOX, while cell safety was determined via MTT and resazurin-based metabolic viability assays. Additionally, a scratch assay was performed on HaCaT keratinocytes to assess the compounds’ effects on cell migration and wound closure.
Key Findings
• Potent Anti-Inflammatory Action: Both SA H and YA B significantly reduced the release of pro-inflammatory cytokines—specifically IL-1β, IL-6, and TNF-α—from PBMCs and skin cells with an efficacy often comparable to or exceeding that of rosmarinic acid.
• Inflammasome Inhibition: SA H and YA B were identified as efficient inhibitors of NLRP3 inflammasome formation, as evidenced by a 43–60% reduction in ASC speck generation in reporter cells.
• Strong Enzyme Inhibition: SA H demonstrated significant inhibitory effects on 5-lipoxygenase (5-LOX) with an IC50 of 2.41 µg/mL and cyclooxygenase-2 (COX-2) with an IC50 of 11.53 µg/mL.
• Supportive Wound Closure: At a concentration of 50 µg/mL, SA H showed a statistically significant increase in the migration of keratinocytes, suggesting it can mildly support the physical closure of wounds.
• Cellular Safety and Hemocompatibility: None of the examined acids exhibited cytotoxicity toward skin cells or PBMCs. Furthermore, they were generally hemocompatible, although SA H and RA displayed a minor procoagulant effect at the highest tested concentration (50 µg/mL).
This research is the first to comparatively demonstrate the therapeutic potential of SA H and YA B as advanced anti-inflammatory agents for wound healing. The novelty of this research lies in identifying that these RA-derived oligomers can modulate the inflammatory response across multiple molecular levels, including the direct inhibition of eicosanoid-producing enzymes and the suppression of the NLRP3 inflammasome. The future implications of these findings suggest that SA H and YA B could be developed into novel topical pharmaceuticals or cosmeceuticals designed to treat chronic inflammatory skin conditions and accelerate healing. Future studies will be required to confirm these effects in in vivo models and to further investigate the long-term safety and optimal formulation strategies for clinical application.
Link to the study: https://www.mdpi.com/1420-3049/31/3/452#