Amaranth (R2) is a synthetic, water-soluble azo dye frequently used as a colorant in cosmetics and food products. Despite its utility, safety concerns persist because the reductive cleavage of azo bonds in such dyes can release aromatic amines, some of which are known carcinogens. Consequently, R2 is strictly regulated; for instance, it is prohibited in children’s products in Korea and banned as a food additive in the United States. To ensure consumer safety, regulators rely on risk assessments that require a Systemic Exposure Dose (SED) calculation, which is impossible without accurate dermal absorption data. This study addresses the existing data gap by using the Franz diffusion cell system to evaluate the percutaneous absorption of R2, providing a scientific basis for its continued use or substitution in cosmetic formulations.
Methods
The researchers evaluated the dermal absorption of 1% Amaranth in skin lotion and cream formulations applied to excised rat dorsal skin for 24 hours. Quantitative analysis was conducted using a validated HPLC method to measure R2 levels in the receptor fluid, skin wash, stratum corneum, and viable skin. All procedures followed OECD Guideline 428 and Korea Ministry of Food and Drug Safety standards to ensure methodological reliability.
Key Findings
• Method Validation: The HPLC analytical method showed excellent linearity (r² > 0.9996), with accuracy and precision levels consistently within regulatory limits for all skin matrices.
• Formulation-Dependent Absorption: The total dermal absorption was minimal, recorded at 3.4 ± 2.7% for skin lotion and 0% for cream.
• Surface Retention: Most of the dye remained on the skin surface and was easily removed, with wash-out recoveries of 76.1% for lotion and 91.0% for cream.
• Systemic Absence: Notably, no R2 was detected in the receptor fluid for either formulation, suggesting it is unlikely to enter systemic circulation.
• Physicochemical Correlation: The observed low absorption is consistent with R2’s high molecular weight (604.5 Da) and low LogP (-5.13), both of which are markers for limited skin penetration.
This research is novel as it provides the first validated dermal absorption data for Amaranth in cosmetic vehicles. By demonstrating that absorption is negligible even in a high-permeability rat skin model—often considered a “worst-case scenario”—the study reinforces that systemic risk from R2 in cosmetics is extremely low. Future implications of this work include providing a robust data set for regulatory risk assessments globally; however, future research should aim to confirm these results using human-relevant models and a broader range of cosmetic excipients to further refine safety margins.
Link to the study: https://www.mdpi.com/2079-9284/13/2/50
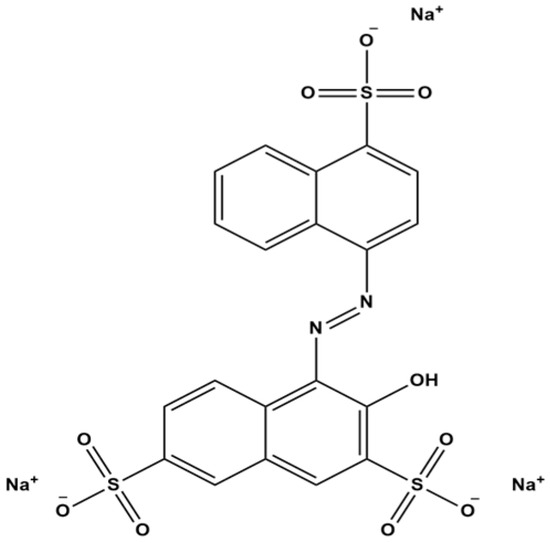
In the image: Chemical structure of Amaranth (R2).
