Epidermal Growth Factor (EGF) is a critical protein for skin health, promoting collagen synthesis, wound healing, and anti-aging through the stimulation of cell proliferation. However, its topical application is significantly hindered by its hydrophilic nature and large molecular size, which result in poor penetration of the stratum corneum and high susceptibility to proteolytic degradation. To overcome these stability and delivery challenges, microencapsulation by complex coacervation (MCC) using Arabic gum and gelatine (AG/GE) was proposed. This solution was selected because it utilizes biocompatible polymers and mild aqueous processing conditions to protect sensitive bioactives while providing a scalable delivery system for cosmetic formulations.
Methods
EGF was encapsulated by combining the protein with a gelatine solution and an oil phase through emulsification, followed by the addition of Arabic gum. Coacervation was triggered by adjusting the pH from 6.3 to 3.9, after which the microcapsules were crosslinked using EDC/NHS and concentrated via phase separation. The resulting 0.3% microcapsule dispersion was incorporated into a minimalist hydrating cream for physicochemical characterization. Finally, the formulation’s safety and efficacy were evaluated through dermatological irritation tests and a 60-day performance study involving twenty human volunteers.
Key Findings
- The process achieved an encapsulation efficiency of 78.8 ± 1.0%, producing spherical, multinucleated microcapsules.
- Dermatological testing classified the formulation as non-irritant, with a Mean Irritation Index (M.I.I.) ≤ 0.2.
- Volunteer assessments showed a 31% reduction in surface spots, a 21% reduction in brown spots, and a 10% reduction in pore visibility.
- Skin texture improved by 22%, and elasticity (measured by retraction time) increased by 33%.
- The formulation significantly strengthened the skin barrier, resulting in a 25% decrease in transepidermal water loss (TEWL).
The novelty of this research is the successful extension of the AG/GE microencapsulation platform—previously limited to lipophilic compounds—to a hydrophilic macromolecule like EGF. This achievement demonstrates a highly versatile delivery architecture that can accommodate diverse classes of bioactives. Future implications suggest the potential for creating advanced skincare products that simultaneously deliver multiple bioactive types within a single system. However, the researchers emphasize that these preliminary results require validation through larger-scale clinical studies to establish greater statistical robustness.
Link to the study: https://www.mdpi.com/2079-9284/13/2/89
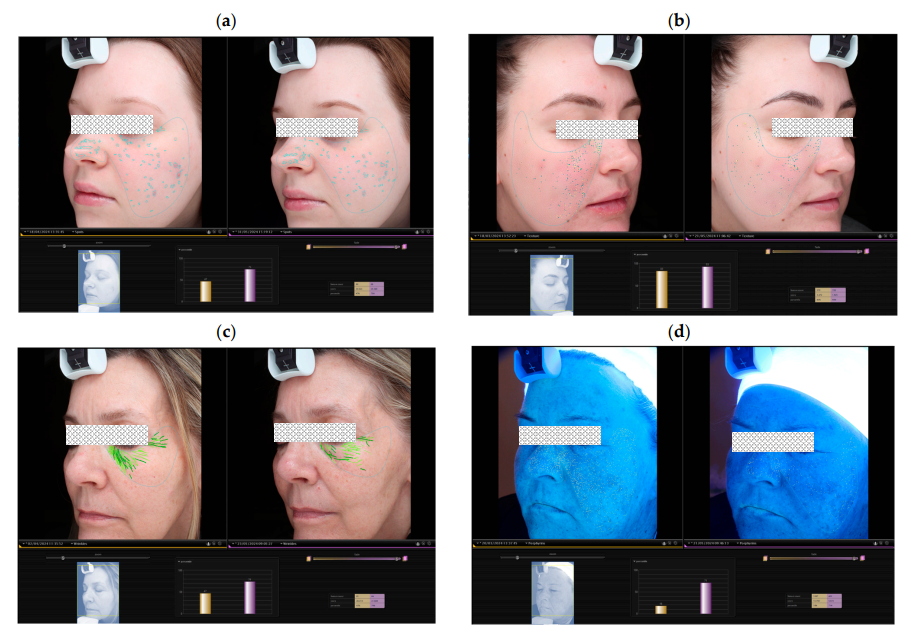
In the figure: Examples of skin measurements performed over the 30 and 60-day study period, evaluating (a) spots and (b) texture in volunteers using MC EGF and (c) wrinkles and (d) brown spots in
volunteers using commercial formulation (CF). Baseline and post-treatment measurements are represented in yellow and purple bars, respectively.
