Stem cells have become central to modern regenerative medicine and dermatology due to their unique abilities to self-renew and differentiate into diverse specialized cell types. However, the use of certain stem cell types can be hindered by ethical concerns, while direct cell transplantation presents complex clinical challenges. Human amniotic mesenchymal stem cells (hAMSCs) offer a powerful alternative as they are multipotent, exhibit low immunogenicity, and avoid ethical issues associated with embryonic sources. Rather than transplanting the cells themselves, researchers have turned to human stem cell-derived conditioned media (hSCCM), which captures the rich mixture of cytokines, growth factors, and extracellular vesicles secreted by these cells to support tissue repair and anti-inflammatory processes topically.
Methods
The researchers characterized hAMSCs using flow cytometry for specific MSC biomarkers and identified the proteomic composition of hSCCM through LC-MS/MS. Biological activity was quantified via dermal fibroblast proliferation assays to evaluate concentration-response, shelf life (up to 29 months), and temperature stability (4 °C to 50 °C). A murine wound model was utilized for exploratory histological assessment of follicular regeneration. Finally, a 10% hSCCM-enriched lotion underwent microbial and physical stability testing, alongside a single-user retrospective observational analysis.
Key Findings
- Stem Cell Identity and Viability: Flow cytometry confirmed that the starting hAMSCs expressed defining MSC biomarkers (CD73, CD105, and CD90) with high viability exceeding 96%.
- Threshold for Biological Activity: hSCCM demonstrated a significant dose-dependent increase in fibroblast proliferation at concentrations of 5% or higher, with a maximum effect noted at 20%.
- Stability and Shelf Life: The biological activity of the conditioned media remained robust for up to 29 months of storage and was unaffected by acute temperature fluctuations between 4 °C and 50 °C.
- Enhanced Proteomic Profile: hSCCM contained 1547 unique proteins, a 56.8% increase over control media, including critical skin-repair factors like CCN1, TGF-β1, fibronectin, and laminins.
- Regenerative Potential: Treated murine wounds showed a substantial trend toward improved follicular regeneration compared to controls, suggesting a shift toward functional tissue repair.
- Cosmetic Formulation Success: The hSCCM-enriched lotion met early-stage microbial safety standards and maintained physical stability under moderate thermal and mechanical stress.
The novelty of this research lies in its comprehensive preclinical characterization of an amniotic-derived secretome as a sterile, cell-free bioactive ingredient that retains functional potency over extended periods. Unlike traditional cosmetics, this formulation leverages a highly interconnected, non-random network of human-derived proteins specifically aligned with extracellular matrix (ECM) organization and angiogenesis. The future implications of these findings suggest a broad translational potential for hSCCM in advanced aesthetic dermatology and integumentary care, providing a scientifically grounded alternative for non-invasive skin rejuvenation and tissue maintenance.
Link to the study: https://www.mdpi.com/2079-9284/13/2/91
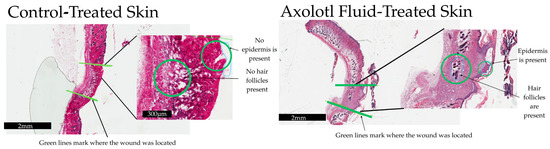
In the figure: Follicular count of control- and hSCCM (Axolotl Fluid)-treated wounds. Green lines mark where the wound was located, and images have been expanded. Left (control) shows no hair follicles or epidermis present on the wound site. Right (hSCCM) has hair follicles and epidermis present where the wound site was located. Scale bars for the low-magnification image are 2 mm and 300 µm for the high-magnification im-age. Images created in Canva (Canva Pty Ltd., Sydney, Australia).
