Cutaneous photoaging involves visible manifestations like wrinkles and texture alterations that significantly impact an individual’s psychosocial well-being, often leading to decreased self-esteem and social anxiety. While retinoids, particularly tretinoin, are considered the cornerstone of anti-aging therapy due to their ability to stimulate collagen and regulate cell turnover, their clinical utility is frequently hampered by “retinoid dermatitis,” characterized by erythema, scaling, and burning. This challenge necessitates the development of advanced formulations that can deliver high efficacy while improving patient compliance through better tolerability. The potential solution evaluated in this research is a novel retinol formulation (ASM® Retinol Texture Repair Emulsion), which utilizes a specialized delivery system designed to enhance stability and cutaneous penetration while hypothesizing a significant reduction in local irritation.
Methods
This prospective, rater-blinded pilot study involved 21 women with moderate-to-severe photoaging who followed a 180-day “step-up” application protocol, gradually increasing from three nights a week to nightly use. Clinical efficacy was quantified using the validated photonumeric Rao–Goldman scale and VISIA® multi-spectral image analysis, with a primary endpoint of achieving a Grade 2 (fine wrinkles) classification. Safety was monitored through dermatological evaluations of adverse events, such as erythema and desquamation, at scheduled intervals including a telematic 120-day assessment.
Key Findings
- Significant Clinical Improvement: By day 180, 76.2% of participants transitioned to the “fine wrinkle” category (Grade 2) on the Rao–Goldman scale, a statistically significant improvement from baseline (p = 0.028).
- High Global Success Rate: Investigator assessments documented “significant” or “marked” global improvement in 85% of the study population.
- Rapid Patient Satisfaction: Unlike the investigator’s timeline, 80% of patients perceived benefits as early as day 30, with a strong final correlation (kappa = 0.840) between patient and clinician assessments.
- Successful Retinization: While 81% of subjects initially experienced mild-to-moderate adverse effects like peeling and redness, these decreased to 23.8% by the study’s end, confirming the skin’s successful adaptation to the molecule.
- Instrumental Trends: VISIA® analysis showed numerical reductions in wrinkle and texture scores, though these trends did not reach statistical significance, likely due to the small pilot sample size.
The novelty of this research lies in its successful validation of a specialized retinol delivery system that balances high-potency efficacy with a manageable safety profile, facilitated by a strict “step-up” dosing protocol that eliminated study dropouts. Furthermore, the study highlights the importance of using standardized photonumeric instruments like the Rao–Goldman scale to provide more granular, age-independent assessments of epidermal remodeling than traditional scales. Future implications include the need for larger-scale randomized controlled trials and histological analyses to directly quantify collagen neo-collagenesis, which will further establish this formulation as a primary non-invasive intervention for long-term photoaging management.
Link to the study: https://www.mdpi.com/2079-9284/13/2/95
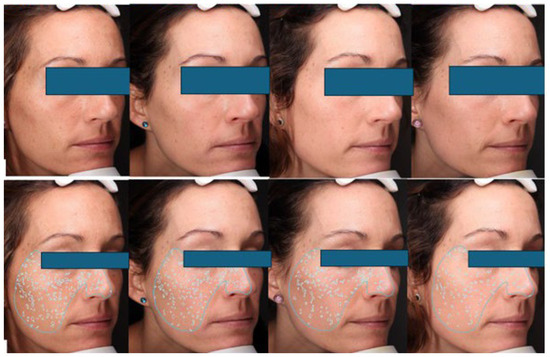
In the figure: VISIA pictures are standardized digital photographs. Upper row: The patient who shows improvement in texture and tone during the treatment, according to both blind raters. Botton row: Patient showing dark spot improvement according to both blind raters.
