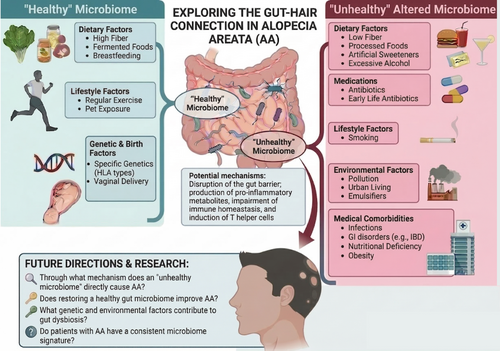
Alopecia areata (AA) is a T-cell-mediated autoimmune disease characterized by hair loss, and researchers are increasingly investigating the gut microbiome as a potential therapeutic target. The gut microbiome consists of trillions of microorganisms that play a critical role in developing systemic immune tolerance and regulating effector T-cell responses. The proposed solution of targeting the microbiome is based on the “gut-immune-skin axis,” where microbial metabolites, such as short-chain fatty acids (SCFAs), help maintain immune homeostasis and may influence receptors at distant sites like the skin. Consequently, gut dysbiosis (imbalance) is hypothesized to weaken the epithelial barrier and shift immune signaling toward pro-inflammatory pathways, potentially increasing susceptibility to AA.
Methods
This review article is a viewpoint analysis based exclusively on a review of published literature regarding the relationship between the gut microbiome and AA. The authors examined evidence from approximately a dozen small, primarily cross-sectional studies, as well as anecdotal case reports and limited randomized clinical trials. The methodology focused on addressing four key uncertainties: the consistency of microbiome signatures, the causal role of dysbiosis, the timing of microbial shifts, and the efficacy of restorative interventions.
Key Findings
- Inconsistent Microbiome Signatures: Small cross-sectional studies have identified differences in gut microbiota, but findings are inconsistent; for example, certain bacterial taxa are reported as both increased and decreased across different studies.
- Lack of Reproducibility: Efforts to identify a reproducible “microbiome signature” for AA have largely failed, and Mendelian randomization studies show little overlap in microbial signals.
- Uncertain Causality: It is currently unknown whether gut dysbiosis is a cause of AA, a consequence of systemic inflammation, or merely an epiphenomenon reflecting environmental factors like diet.
- Limited Clinical Evidence for Treatments: Interventions such as probiotics and the Mediterranean diet have not shown significant clinical benefits for AA in controlled studies.
- Anecdotal Fecal Microbiota Transplant (FMT) Results: While some case reports show hair regrowth after FMT, these instances are often linked to the treatment of unrelated infections (like C. difficile), and spontaneous regrowth cannot be ruled out.
The novelty of this research lies in its critical confrontation of “clinical excitement” with the reality of current scientific evidence, highlighting that the hype surrounding microbiome therapies is currently outpacing proven results. The sources conclude that there is insufficient evidence to recommend microbiome-directed testing or therapies to AA patients at this time. For future implications, the research underscores the urgent need for rigorous, longitudinal, and prospective studies in diverse populations to establish whether a causal link exists and if restoring the microbiome can lead to sustained clinical improvements.
Link to the study: https://onlinelibrary.wiley.com/doi/full/10.1111/ijd.70437