Human skin pigmentation is primarily determined by melanin production, a process catalyzed by the enzyme tyrosinase. While melanin protects against UV radiation, its overproduction leads to hyperpigmentation and dark spots, creating a demand for safe, natural skin-brightening agents. Traditionally, plants like Equisetum ramosissimum (horsetail) have shown promise due to their rich phytochemical content, but conventional extraction methods often require large quantities of toxic solvents that pose risks to human health and the environment. To address this, Microwave-Assisted Extraction (MAE) was investigated as a “green” alternative because it reduces solvent consumption and extraction time while protecting heat-sensitive bioactive compounds. This study seeks to optimize MAE conditions using ethanol, a safer solvent, to maximize the recovery of anti-tyrosinase compounds from E. ramosissimum.
Methods
Ground E. ramosissimum powder was extracted with 95% ethanol using MAE under varying temperatures (40–90 °C), durations (15–45 min), and solvent ratios (1:8–1:16). The extracts were subsequently analyzed for total phenolic content, antioxidant activity, and in vitro tyrosinase inhibition. Key bioactive compounds were identified through LC-MS/MS analysis. Finally, molecular docking simulations were performed to evaluate the binding affinities and mechanisms of these compounds against both mushroom and human tyrosinase.
Key Findings
- Optimal Extraction Conditions: The strongest anti-tyrosinase activity (IC50: 126.48 ± 6.668 µg/mL) was achieved using a solid-to-solvent ratio of 1:16 at 40 °C for 15 minutes (sample MCW5).
- Temperature Effects: While higher temperatures (90 °C) increased the total phenolic content and antioxidant activity, they diminished anti-tyrosinase efficacy, likely due to the degradation of heat-sensitive inhibitory compounds.
- Identification of Bioactive Markers: LC-MS/MS identified kaempferol-3-gentiobioside (KF-3-GBS) as a unique marker in the most active extract, which was not reported in previous studies using traditional maceration.
- Molecular Docking Insights: Resveratrol isomers, KF-3-GBS, and 1-16:0-lysoPE demonstrated strong predicted binding to human tyrosinase.
- Inhibitory Mechanism: Resveratrol isomers were found to interact directly with copper ions in the enzyme’s active site, suggesting a copper-chelation mechanism for tyrosinase inhibition.
The novelty of this research lies in establishing an energy-efficient and sustainable MAE protocol specifically tailored to preserve the anti-tyrosinase compounds of E. ramosissimum. Furthermore, it provides the first detailed molecular docking analysis of this plant’s constituents against human tyrosinase, identifying kaempferol-3-gentiobioside as a primary candidate for direct enzyme inhibition. The future implications of this study include the potential use of these extracts as natural active ingredients in skin-brightening cosmetics. Future work will focus on validating these findings in cellular models and ensuring the stability of the extract in final cosmetic formulations.
Link to the study: https://www.mdpi.com/2079-9284/13/3/107
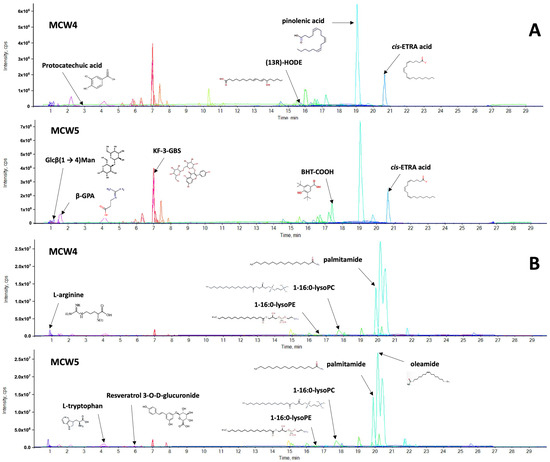
In the figure: The key compounds chromatograms of E. ramosissimum crude extracts (MCW4 and MCW5). (A) negative ion mode and (B) positive ion mode.
