Acne vulgaris is a chronic inflammatory disease affecting nearly 10% of the global population, with a prevalence of up to 85% among adolescents. Beyond physical lesions, it imposes a heavy psychosocial burden, often leading to anxiety, depression, and reduced quality of life. While topical therapies like antibiotics and benzoyl peroxide are mainstays, they are limited by rising antimicrobial resistance and frequent side effects such as irritation and erythema. Specifically, the irritation from benzoyl peroxide is attributed to its degradation into benzoic acid. Stabilized 1% hydrogen peroxide (H2O2) was considered a potential solution because it acts as a reactive oxygen species (ROS) with antibacterial and debriding properties while maintaining a more favorable tolerability profile compared to traditional treatments.
Methods
This work comprised two open-label, prospective clinical studies involving 42 participants with facial acne and 41 with truncal acne, who applied novel stabilized 1% H2O2 formulations twice daily for 8 weeks. The facial group used a cream-gel, while the truncal group utilized a sprayable lotion designed for large and hard-to-reach areas. Researchers evaluated efficacy through clinical scales (IGA, EGAE), lesion counts, and various biometric measurements, including sebum production, porphyrin fluorescence, and skin pH.
Key Findings
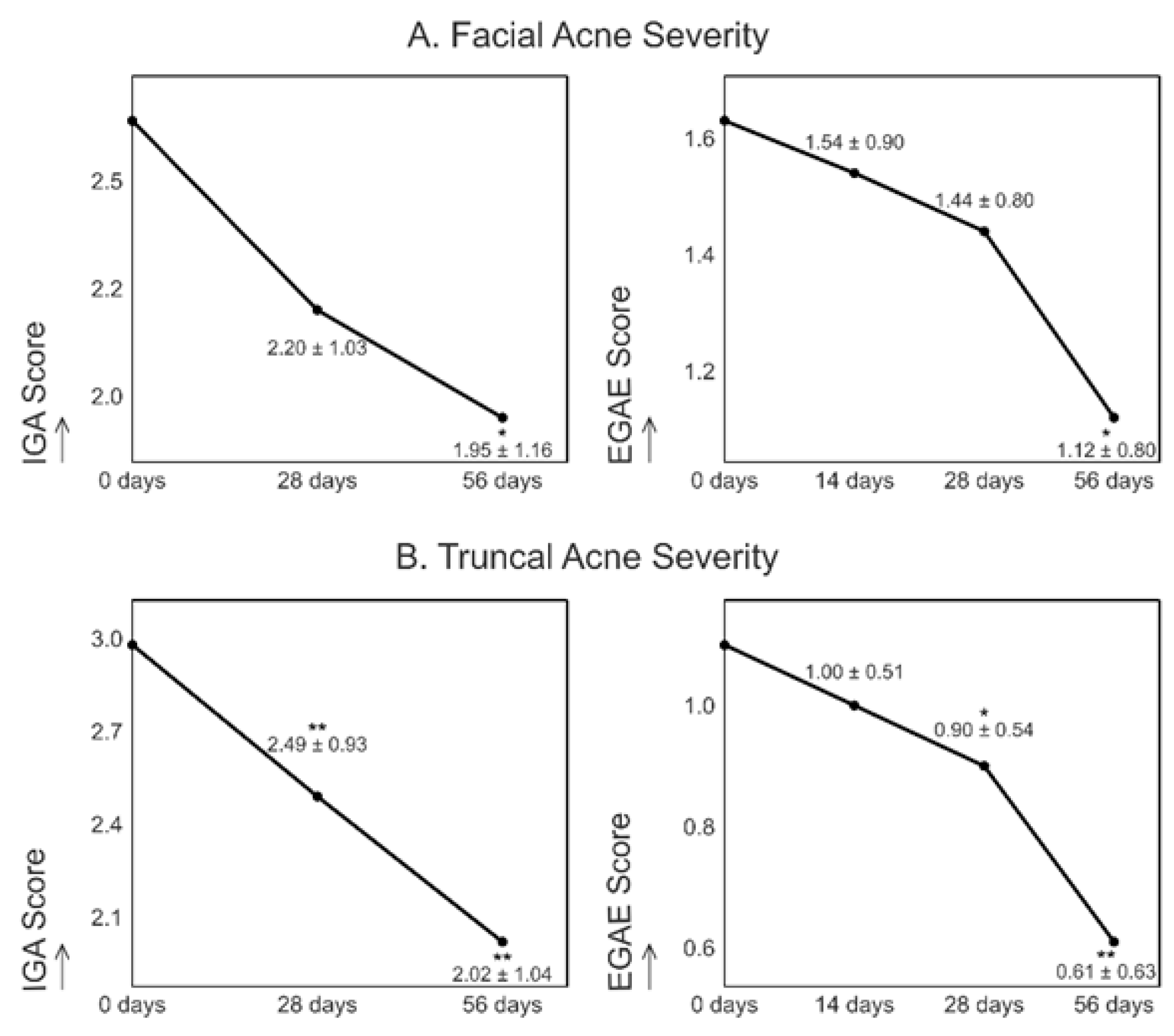
- Clinical Improvement: By day 56, facial acne severity decreased by 26% (IGA) and 31% (EGAE), while truncal acne severity improved by 32% (IGA) and 45% (EGAE).
- Lesion Reduction: Facial papules were reduced by 45%, and truncal inflammatory lesions showed a significant overall reduction of 60%.
- Biometric Effects: Sebum production on the face decreased by 75% by the end of the study. Significant reductions in porphyrin count and size were observed, indicating decreased C. acnes virulence.
- Skin Health: Both facial and truncal acne showed significant improvements in erythema (35% and 7% reductions, respectively) and skin desquamation, with some improvements noted as early as two hours after application.
- Patient Satisfaction and Safety: The formulations were highly accepted, with 83% to 95% of participants reporting satisfaction. Unlike benzoyl peroxide, these formulations caused no clothing or bed linen discoloration and had minimal local irritation.
The novelty of this research lies in the development and evaluation of specific stabilized 1% H2O2 formulations—particularly the sprayable format for truncal acnew hich address the practical and physiological challenges of treating extensive body areas. These formulations demonstrate a multifactorial mechanism of action that targets inflammation, bacterial virulence, and sebum production without the irritation typically associated with benzoic acid. Future implications suggest that these 1% stabilized H2O2 options could be integrated into routine clinical practice as patient-friendly, effective alternatives to traditional therapies, though further randomized, controlled studies are needed to confirm these preliminary outcomes in larger populations.
Link to the study: https://www.mdpi.com/2079-9284/13/2/52
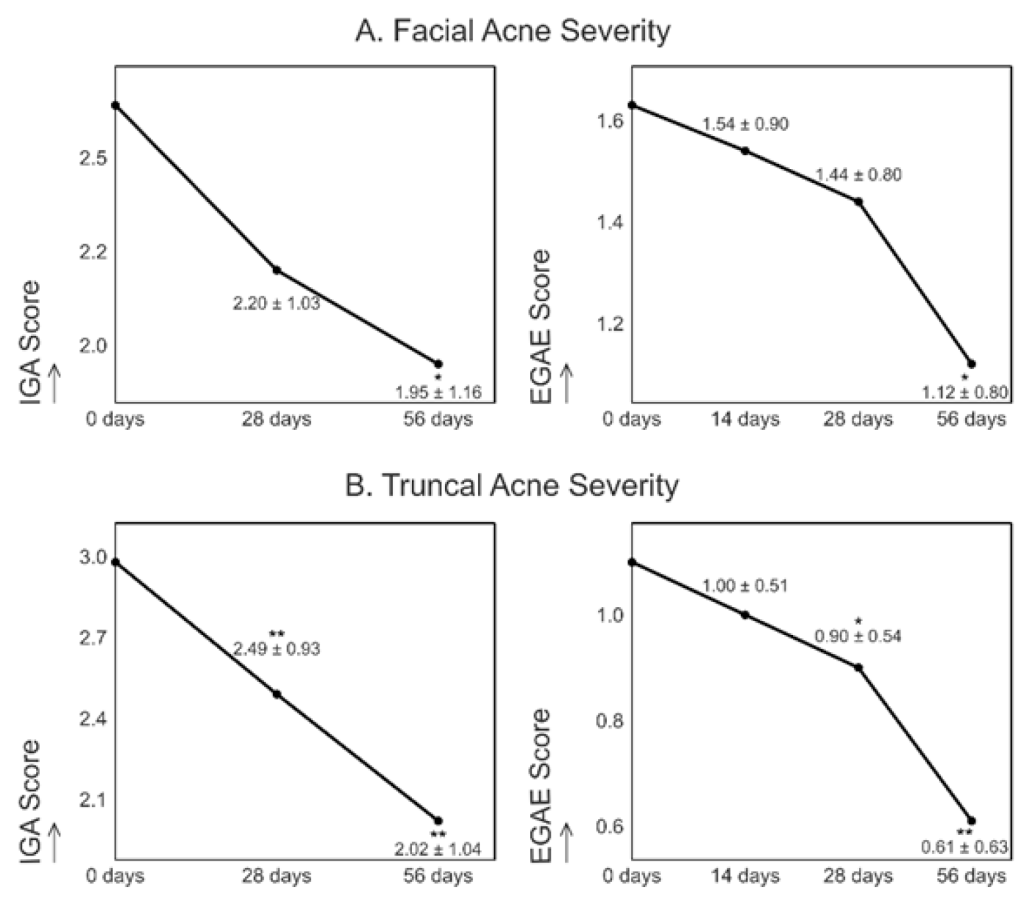
In the image: Evolution of facial (A) and truncal (B) acne severity measured by IGA or EGAE score over the course of the study, expressed as mean ± standard deviation (SD) at different time points. * p < 0.05 versus baseline (mean ± SD). ** p < 0.01 versus baseline (mean ± SD).