The traditional scientific focus on gene therapies and genetic variants as the primary drivers of health and disease has often overlooked the critical role of proteins and epigenetics. Research indicates that “proteostasis collapse”—the accumulation of damaged, misfolded, or oxidized proteins—is a major cause of aging and skin degradation, rather than DNA mutations alone. For example, long-lived proteins like collagen accumulate damage over decades, leading to “fibroblast collapse” and a loss of regenerative capacity in the skin. To address this, a “systems-therapeutic” approach using the whole secretome from both adipose mesenchymal stem cells (ADSCs) and fibroblasts (FBs) was considered. This combination was chosen because the complete secretome, including both the soluble fraction and extracellular vesicles, provides a more comprehensive set of signaling mechanisms to reduce inflammation and elicit a regenerative state compared to reductionist treatments like isolated exosomes.
Methods
Adipose mesenchymal stem cells and fibroblasts were cultured under hypoxic conditions to optimize their regenerative potential. The whole conditioned media was collected separately for each cell type and subjected to protein digestion using a standardized protocol. Unbiased relative quantification of total protein levels was performed using the TMTpro™ MS3 workflow on an Orbitrap Ascend™ Tribrid™ mass spectrometer. Finally, the acquired spectra were searched against a database of human protein sequences to characterize the secretome’s components.
Key Findings
- Pathway Diversity: The combined secretome engages at least 16 major therapeutic pathways comprising hundreds of signaling mechanisms to benefit various skin conditions.
- Structural Focus: The most significantly up-regulated proteins across both cell types are primarily involved in collagen fibril organization and skin development.
- Growth Regulation: Key pathways identified include extracellular matrix (ECM) organization and IGF regulation, which are essential for fibroblast function and maintaining youthful skin.
- Functional Categories: The analysis revealed a rich spectrum of proteins, including:
- Immune Modulators: Proteins that reduce inflammation and bias the immune system toward a regenerative M2 macrophage phenotype.
- Senomorphics: Molecules such as Calmodulin-like protein 5 and FAF1 that suppress cellular senescence without destroying the cells.
- Antioxidants: A “redox hub” of natural antioxidants like Thioredoxin and Catalase that work synergistically to neutralize free radicals.
- Chaperones/Heat Shock Proteins: Proteins that protect, repair, or recycle misfolded proteins to maintain cellular health.
- ECM Maintenance: The secretome contains proteins like dermatopontin and various collagen types that rebuild the dermal matrix and basement membrane without causing fibrosis.
The novelty of this research lies in its shift toward “proteome renormalization” as a therapeutic strategy. By utilizing the whole secretome rather than isolated components, the study provides a blueprint for a systems-level intervention that restores a youthful proteomic environment to aged or damaged skin. This approach addresses the complexity of skin health by simultaneously targeting inflammation, senescence, and structural decay. The future implications of this work are significant, suggesting that multiomic studies—incorporating lipids, peptides, and non-coding RNA—will be essential to fully unlocking the potential of stem cell-based therapies for a wide array of dermatological and systemic conditions.
Link to the study: https://www.preprints.org/frontend/manuscript/291cc3e9d853f4a2f9139d2ea41709b8/download_pub
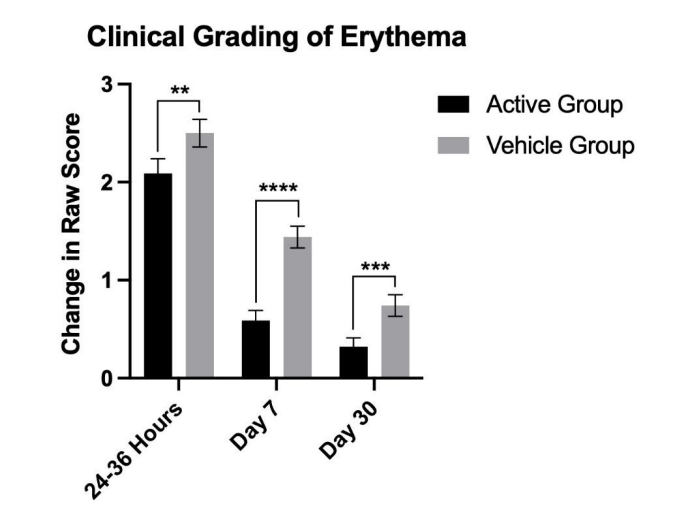
Figure 1: From Ngyuen et al. (2026). NeoGenesis Recovery, containing the same ADSC and FB secretome as used
in this study, significantly (statistically and clinically) reduces erythema and inflammation for the duration of a
30-day study.
